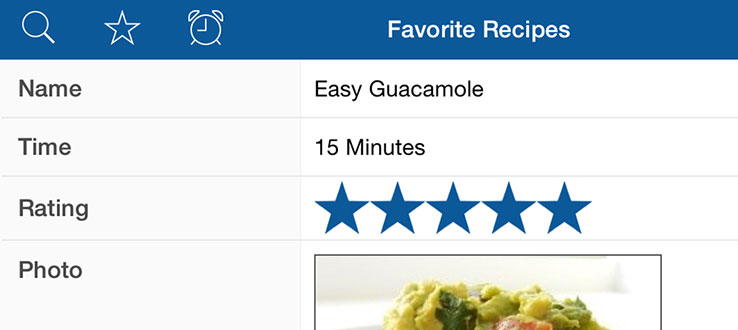
Therefore, we evolved in a detailed screen a solubilization, purification, and reconstitution procedure for heterologously expressed TAP. Interestingly, CL1 and CL2 of TAP1 interact with the X-loop of TAP2 as shown by cysteine cross-linking ( 24).įor advanced biochemical and biophysical analyses of the molecular mechanism of TAP, it is essential to work with purified protein. A short stretch of the cytosolic loop 1 (CL1) of TAP1 is involved in the communication between the NBD and TMD as identified by a chemical protease ( 23). 
However, it is not solved yet whether TAP has a basal ATPase activity as found by other ABC transporters. Although ATP binding to TAP is peptide-independent ( 20), ATP hydrolysis is tightly coupled to peptide binding and translocation ( 21, 22). From biochemical and structural studies with soluble NBDs as well as fully assembled ABC transporter complexes, it has become clear that ATP binding induces a rigid body movement of the helical domain toward the catalytic domain, which then allows dimerization of the NBDs in a head-to-tail orientation ( 17, – 19). Peptide binding is a two-step process with a fast association followed by a slow structural rearrangement comprising 25% of TAP residues ( 11, 12).Įach NBD is divided into a helical and catalytic domain and contains the Walker A/B motifs and C-loop (ABC signature) sequence, all of which are involved in the binding and hydrolysis of ATP ( 13, – 16). However, it is still an open issue whether only one or several peptides bind to TAP with high affinity at the same time. Peptide binding is ATP-independent and follows a Langmuir (1:1) isotherm ( 7, 11). TAP shows a preference for protein fragments of 8–16 residues ( 7) with a specificity restricted to the three N-terminal and the C-terminal residues ( 8, – 10). Both TMDs together form the translocation pathway, with the peptide-binding site located at cytosolic loops ( 6). Each of the subunits is composed of a transmembrane domain (TMD) and a C-terminal cytosolic nucleotide-binding domain (NBD). TAP forms a heterodimer of the half-transporters TAP1 and TAP2. TAP belongs to the family of ABC transporters, which translocate a very large range of solutes across membranes under consumption of ATP ( 3, – 5). Viruses and tumor cells have therefore evolved strategies to shut down antigen presentation, for example by down-regulation or inhibition of TAP (for review see Refs. Recognition of an antigenic peptide in complex with MHC class I by a specific T-cell receptor induces the elimination of the infected or malignantly transformed cell. Kinetically stable peptide-MHC complexes traffic to the cell surface, where they are scanned by cytotoxic T-lymphocytes. TAP translocates proteasomal degradation products from the cytosol into the endoplasmic reticulum lumen, where these peptides can bind to major histocompatibility complex (MHC) class I molecules. 
The transporter associated with antigen processing (TAP) 5 has a key role in the adaptive immune system of higher vertebrates. These results represent an optimal starting point for detailed mechanistic studies of the transport cycle of TAP by single molecule experiments to analyze single steps of peptide translocation and the stoichiometry between peptide transport and ATP hydrolysis. In addition, the TAP complex shows strict coupling between peptide binding and ATP hydrolysis, revealing no basal ATPase activity in the absence of peptides. This allowed us to determine the substrate-binding stoichiometry of the TAP complex by fluorescence cross-correlation spectroscopy. To examine this ABC transport complex in mechanistic detail, we have established, after extensive screening and optimization, the solubilization, purification, and reconstitution for TAP to preserve its function in each step. 
The transporter associated with antigen processing (TAP) is an essential machine of the adaptive immune system that translocates antigenic peptides from the cytosol into the endoplasmic reticulum lumen for loading of major histocompatibility class I molecules.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
